|
Introduction Materials and Methods Results Discussion Acknowledgements Literature Cited |
ABSTRACT: I divided 60 Loggerhead Shrikes into five groups, and starting the day after hatching dosed them with either 0, 1. 2, 4. or 8 mg of dieldrin per kilogram of body weight per day. Mean age at death was 78.25 days for the 1-mg group, 36.75 days for the 2-mg group, 21.33 days for the 4-mg group, and 16.75 days for the 8-mg group. When the shrikes in the 0- and 1-mg groups were 25 days old, I began presenting them a cricket and mouse daily and watched their behavior.
The two groups showed no significant difference in their ontogeny of cricket killing, but the ontogeny of mouse killing was significantly prolonged in the treated birds. This study is the first to determine the chronic toxicity of an organochloricle insecticide to an insectivorous passerine, and the first to demonstrate an insecticide-induced change in a complex behavioral ontogeny.
San Diego State University, San Diego, California 92115. Present address: Section of Ecology and Systematics, Ithaca, New York 14853. Accepted 16 May 1975.
![]() ost studies on the toxicity of the commonly-used organochloride insecticide dieldrin to birds have been on such seed-eating species as doves, quail, pheasants, and ducks (e.g. see Genelly and Rudd 1956, Heath and Stickel 1965, Tucker and Crabtree 1970). Only one study (Basson 1971) investigated the chronic toxicity (toxicity of repeated daily doses) of dieldrin to a passerine, and it was also on a seed-eating bird, the Cape Sparrow (Passer melanurus) of South Africa. A study by Jefferies and Davis (1968) involved the insectivorous Song Thrush (Turdus ericetorum), but it was directed mainly toward the pharmacodynamics of dieldrin, and only one bird died or showed symptoms of poisoning. Data for the toxicity of dieldrin to an insectivorous passerine are needed because data for one type of bird may be meaningless for another type of bird, as shown by Tucker and Crabtree (1970). They found that the LD-50 (dosage that killed 50% of a sample) of dieldrin for House Sparrows (Passer domesticus) was about one-eighth of that for Mallards (Anas platyrhynchos) and about five times that for Gray Partridges (Perdix perdix). To provide information on the chronic toxicity of dieldrin to an insectivorous passerine, I conducted a study on the common and easily-raised Loggerhead Shrike (Lanius ludovicianus).
ost studies on the toxicity of the commonly-used organochloride insecticide dieldrin to birds have been on such seed-eating species as doves, quail, pheasants, and ducks (e.g. see Genelly and Rudd 1956, Heath and Stickel 1965, Tucker and Crabtree 1970). Only one study (Basson 1971) investigated the chronic toxicity (toxicity of repeated daily doses) of dieldrin to a passerine, and it was also on a seed-eating bird, the Cape Sparrow (Passer melanurus) of South Africa. A study by Jefferies and Davis (1968) involved the insectivorous Song Thrush (Turdus ericetorum), but it was directed mainly toward the pharmacodynamics of dieldrin, and only one bird died or showed symptoms of poisoning. Data for the toxicity of dieldrin to an insectivorous passerine are needed because data for one type of bird may be meaningless for another type of bird, as shown by Tucker and Crabtree (1970). They found that the LD-50 (dosage that killed 50% of a sample) of dieldrin for House Sparrows (Passer domesticus) was about one-eighth of that for Mallards (Anas platyrhynchos) and about five times that for Gray Partridges (Perdix perdix). To provide information on the chronic toxicity of dieldrin to an insectivorous passerine, I conducted a study on the common and easily-raised Loggerhead Shrike (Lanius ludovicianus).
Besides killing birds, pesticides can alter their behavior, as many examples among vertebrates show. Sublethal doses of dieldrin have caused poorer performance by domestic sheep on both a vigilance task (Sandler et al. 1969) and the relearning of a visual discrimination problem (Van Gelder et al. 1969). Baxter et al. (1969) found that Ring-necked Pheasant chicks (Phasianus colchicus) from dieldrin-dosed hens tended to choose the deep side of a visual cliff, while controls tended to choose the shallow side. In a field study McEwen and Brown (1966) gave single oral sublethal (loses of dieldrin to a portion of the Sharp-tailed Grouse (Pedioecetes phasianellus) on a breeding ground, thereby causing a change in the social hierarchy and terminating effective breeding behavior in some of the treated birds. Graber et al. (1965) concluded that a colony of Red-winged Blackbirds (Agelaius phoeniceus) became quiescent and abandoned partly constructed nests after being contaminated with dieldrin. Sublethal doses of DDT, another organochloricle insecticide, have been shown to affect salinity selection in Gambusia (Hansen 1969) and temperature selection in young Atlantic salmon (Salmo salar) (Ogilvie and Anderson 1965). Sobotka (1971) found that a small dose of DDT increased the exploratory behavior of laboratory mice (Mus musculus). DDT has also prevented brook trout (Salvelinus fontinalis) from learning a simple light-dark choice to avoid an electric shock (Anderson and Peterson 1969).
None of these studies investigated the effect of an insecticide on a complex behavioral ontogeny. Because some pet shrikes that I had raised appeared to have ontogenies for cricket killing and mouse killing that were complex enough to detect any dieldrin-induced changes, I compared those ontogenies in some of the treated birds with those in a control group.
I collected 60 Loggerhead Shrike nestlings from a nonagricultural San Diego, California region bordering the last ten miles of the now intermittent Sari Diego River, and divided them randomly by nest into five equal groups. Four of the groups received 1, 2, 4, or 8 mg of 87% technical-grade dieldrin insecticide dissolved in corn oil per kg of body weight per day, and the other group (controls) received oil only. As a bird gained weight, the regular Substitution of a stronger Solution of dieldrin allowed oil administration to be maintained between 15 and 30 ml per day. The oil was placed deeply into the shrike's mouth with a blunted Hamilton Company microsyringe.
The nestlings were color-banded and then weighed and dosed at their nests until 10 days old. At this age, they were removed and hand-reared until 38 days old, in 40 x 40 x 90-cm cages containing nest-sized cloth-lined bowls, one clutch per cage. The shrikes were handled frequently and kept in an area of high human activity to insure their tameness. Ages at death were recorded throughout the study.
The shrikes' diet consisted of horse-meat, chopped whole rats and hamsters, vitamins and minerals, and supplements of thoroughly crushed mealworms and crickets. The birds were hand-fed as nestlings and fledglings, and at fledging age (about 20 days) were given constant access to food, water, and forked branches through which sharpened nails had been driven for impaling. After fledging, they were given 30 min to 2 hr a day to exercise in an outdoor flight cage. On the 38th day of age they were permanently transferred to a 2.4 x 24 x 5.4-m outdoor cage.
I have described the prey-killing trials elsewhere (Busbee 1976) but will summarize that description here for the convenience of the reader. Beginning on the 25th day of age, the cricket-killing and mouse-killing behavior of the shrikes was observed daily in a 1.8m-cube cage. Each shrike was offered, in a random order, an adult house cricket (Gryllus domesticus) and a young 13- to 16-g gray house mouse (Mus musculus). The shrike behavior was described into a tape recorder, and time measurements were made to the nearest 5 sec with a stopwatch. The basic trial, 2 min long, was extended 2 min in mouse-killing trials if the shrike had attacked the mouse, and then extended an additional 4 min if the shrike was in such control of the mouse (by holding the mouse with its feet or beak or by severely wounding it) that it did not run from the shrike. Cricket-killing trials were ended on the 44th day of age; mouse-killing trials were ended on the 48th day for the control group and the 58th day for the treated birds.
The shrikes' mean ages at death are in Table 1, which shows a strong tendency for the birds to die sooner at higher doses of dieldrin. All but seven of the birds dying were seen in convulsions at least once, and generally several times. These convulsions, lasting about 15 sec to 2 min, first appeared about 2 or 3 days prior to death. Three of the birds never seen in convulsions were the two O-mg birds that died and the only bird missing from its nest, a member of the 1-mg group that disappeared on the 9th day of age. Because this death was so far outside the 60 to 103 day range of the others in the group (it was 5.9 SD from the mean of the other deaths in the group), it was discarded. Besides convulsions, another sign of impending death was a weight loss beginning a few days prior to death.
During the ontogeny of cricket-killing experiment, a typical first successful trial consisted of the shrike approaching the cricket, bowing and fluttering the win toward it, and then capturing it after one or more misses. A typical later trial consisted of an immediate approach and an immediate capture. All approaches were accompanied by a capture. For both groups the mean ages and standard errors of first occurrence of approaching-capturing, bowing, and fluttering were from 37.0 ± 0.84 to 38.1 ± 0.79 days. All shrikes performed these behaviors at least once. Student's t-tests showed no significant differences between the 0- and I-mg groups. For the 0-mg group median approach times (the time between introduction of the prey and the landing of the shrike in the arena) were 20, 5, and 0 sec for the first three approaches, and 0 sec thereafter. For the I-mg group median approach times were 2-5, 5, 0, and 2.5 for the first four trials, and 0 sec thereafter. Because many of the approach times were very short, medians are presented so that trends would not be obscured by a few unusually long approach times. For both groups mean capture times (the time between the shrike's approach and its grasping the cricket in the beak without letting it go in such a state that it hopped away) were initially about 10 to 13 sec, were down to I or 2 sec by the 5th approach, and reached 0 at the 8th approach. No significant differences were apparent between the 0- and I-mg groups in either approach or capture times.
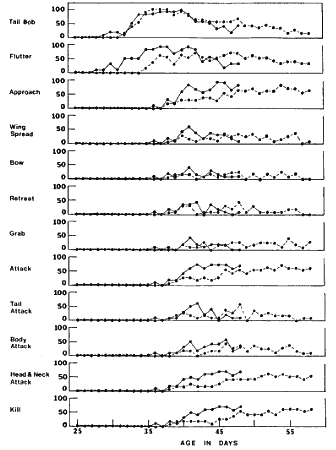
Figure 1. The occurrence of behavior during the ontogeny of mouse killing in young dieldrin-treated and untreated Loggerhead Shrikes that were presented a mouse daily and that killed at least one mouse. The vertical axis represents the percentage of shrikes performing the indicated behavior at least once at tile indicated age. The broken lines represent the 1-mg group, the solid lines the 0-mg group.
| Species | |||
|
Dosage (mg/kg·day |
Loggerhead Shrike (mean ? SE) |
Laughing Dove1 (median) |
Cape Sparrow1 (median) |
| 0.5 | ? | 49.0 | ? |
| 1 | 84.5 ? 3.9 | ? | 85.1 |
| 1.5 | ? | 18.6 | ? |
| 2 | 36.8 ? 2.5 | ? | 34.7 |
| 3 | ? | 8.5 | 20.0 |
| 4 | 21.3 ? 2.1 | 6.8 | ? |
| 6 | ? | ? | 8.1 |
| 8 | 16.8 ? 1.0 | 4.7 | 7.0 |
The behaviors noted during the ontogeny of mouse killing are listed in Fig. 1, together with their occurrence by age of the shrikes. Early trials consisted of the shrike merely looking at the mouse, but tail bobbing and fluttering from the perch soon followed. The first major step, approaching the mouse, was often accompanied by inefficient biting, by grabbing the mouse with the feet, and by wing spreading and fluttering. A comparison of the first days of occurrence for these behaviors is given in Table 2. The entire ontogeny of mouse killing tended to be delayed in the treated group, with fluttering, all forms of attack, grabbing with the feet, and killing occurring significantly later.
Approach times (Fig. 2A) were initially high and then declined steadily. Although four of the 0-mg and three of the 1-mg birds killed on their first approach, kill times (the time between the approach and the death throes of the mouse) were high (Fig. 2B). Kill times also declined steadily, so that a typical later trial consisted of an immediate approach and a quick kill by attacking the neck. No significant differences were apparent between the two groups in either approach or kill times.
For a more detailed account of the ontogenies of cricket killing and mouse killing in the Loggerhead Shrike, see Busbee (1976).
Table 2. Comparison of first ages of occurrence of behavior observed during the ontogeny of mouse killing in deildren-treated and untreated Loggerhead Shrikes
| Dosage (mg/kg�day) | ||||
| 0 | 1 | |||
| Behavior | % of birds performing behavior (n = 10) | Mean age at first occurrence in days ? SE |
% of birds performing behavior (n = 11) | Mean age at first occurrence in days ? SE |
| Tail bob | 100 | 32.7 ? 0.60 | 100 | 33.9 ? 0.39 |
| Flutter | 100 | 32.5 ? 0.901 | 100 | 39.0 ? 1.351 |
| Approach | 90 | 40.3 ? 1.01 | 91 | 43.1 ? 1.40 |
| Wing Spread | 90 | 40.3 ? 1.01 | 91 | 44.0 ? 1.84 |
| Bow | 90 | 40.7 ? 1.00 | 91 | 44.2 ? 1.79 |
| Grab | 60 | 40.3 ? 1.051 | 82 | 47.4 ? 1.781 |
| Attack | 80 | 40.5 ? 1.252 | 82 | 44.6 ? 1.582 |
| Tail attack | 80 | 40.5 ? 1.252 | 82 | 44.8 ? 1.482 |
| Body attack | 80 | 41.1 ? 1.262 | 73 | 45.4 ? 1.532 |
| Head-neck attack | 80 | 40.8 ? 1.322 | 73 | 45.2 ? 1.732 |
| Kill | 80 | 41.0 ? 1.302 | 73 | 46.1 ? 1.902 |
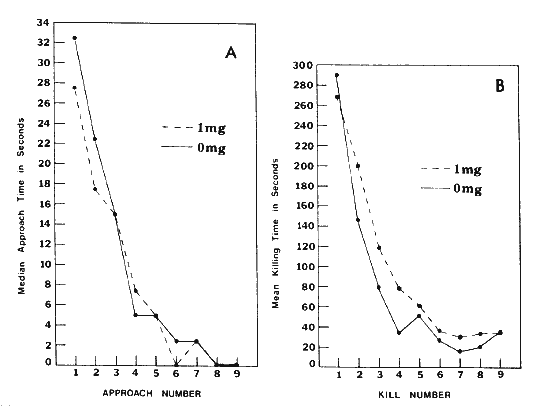
Figure 2. A, Successive median approach times for young dieldrin-treated and untreated Loggerhead Shrikes that were presented a mouse daily. B, successive mean killing times for young dieldrin-treated and untreated Loggerhead Shrikes that were presented a mouse daily and that killed at least three mice.
Mean weights of the untreated group, reported by Busbee (1976), were similar to that of a shrike weighed periodically at its nest by Miller (1931). The weight loss in the treated birds prior to death was similar to that reported by Stickel et al. (1969), who concluded that it resulted from a depletion of flight muscles. No tissues of the dead shrikes were analyzed for dieldrin residues, but convulsions prior to death are typical of organochloride poisoning (Gowdey et al. 1954, Wooley 1969). That death was generally by poisoning is further supported by the absence of convulsions in the two 0-mg birds that died.
Mean ages of the shrikes at death were similar to those of dieldrin-dosed adult Cape Sparrows (Basson 1971) when doses were I or 2 mg/kg·day, but were considerably later when the dose was approximately 8 mg/kg·day (Table 1). (Dosage figures for Basson's study, shown in Table 1, were obtained by dividing median lethal intake of the birds in his three middle dosage groups by the number of days the birds ate, averaging this figure, and then using it to convert the ppm diet figures to mg/kg·day.) An adult Song Thrush fed a 5.33 mg/kg·day dieldrin diet by Jefferies and Davis (1965) died on the 9th day, which agrees well with Basson's (1971) findings on the Cape Sparrow. Although the thrush data were for only one bird, their agreement with the sparrow data, and the agreement between the sparrow data and the shrike data at lower doses suggests that the difference between sparrows and shrikes at higher doses may not be a phylogenetic difference, but may instead be a result of a physiological difference based on age. For example, egg yolk utilization may have some effect on physiological handling of dieldrin. The consistent difference between the shrikes and Laughing Doves (Stigmatopelia senegalensis) is probably phylogenetic, because these doves died in less than half the time required for shrikes to die (Table 1). Other comparisons with the literature are of little value, because dosage regimes differ considerably from that of the present study.
In an unpublished study in which I measured food intake of captive shrikes from their hatching until they were 50 days old, I found they ate approximately 40% of their body weight daily in rat muscle meat, or 10% on a dry-weight basis. Allowing for a 25% increase in food consumption for more active wild shrikes, a single daily dieldrin dosage of 1 mg/kg·day is equivalent to a diet containing 8 ppm on a dry-weight basis, or 2 ppm on a wet-weight basis. A single daily dose may not be the same as a homogeneous diet containing that amount (Kenaga 1973), but problems in mixing dieldrin homogeneously with meat (and its dieldrin-absorbing fat) precluded this method.
These doses may be available in some agricultural regions, as shown in a study by Korschgen (1970). Examining two fields that had been treated for several years with the insecticide aldrin (which is quickly metabolized by animals to dieldrin), Korschgen found the mean wet-weigh t-based dieldrin content of the cricket Gryllus assimilis to be 0.22 ppm, the ground beetle Harpalus pennsylvanicus to be 0.99 ppm, and the ground beetle Poecilus chalcites to be 9.33 ppm. During weather conditions favoring soil retention of aldrin and dieldrin, dieldrin content reached 36.2 ppm for P. chalcitcs, 4.60 ppm for the toad Bufo americanus, 12.2 5 ppm for the garter snake Thamnophis sirtalis, and 0.96 ppm for the white-footed mouse Peromyscus maniculatus.
The shrikes I treated with a 2-ppm diet all died within 103 days of age. That this dosage could be obtained by shrikes feeding in the fields examined by Korschgen (1970) is indicated by Miller's (1931) estimate of shrike diet components: Beetles comprise 20% of the diet, crickets and grasshoppers from 30 to 70%, small mammals 3 to 55%, reptiles probably over 8%, and amphibians an unknown percentage, but toads are eaten.
The results show dieldrin-treated shrikes beginning several steps of their mouse killing ontogeny later than untreated shrikes. Behavioral changes induced by pesticides have little practical importance if they occur only in animals already doomed to die of physiological disruptions caused by those pesticides. Therefore the delayed ontogeny of mouse killing is of practical importance only if the shrikes would have lived had dieldrin dosage been stopped after the Behavorial study. The need for toxicity data precluded cessation of dosing, so it must be determined in retrospect whether the birds would have survived had the dosing been stopped. Tucker and Crabtree (1970), after a study involving acute doses of dieldrin in nine bird species including the House Sparrow, concluded that mortalities usually occurred 1 to 9 days following treatment. Although dynamics of acute and chronic poisoning differ, the fact that the shrikes in the present study died an average of 38 days after the mean day of first kill (over four times Tucker and Crabtree's usual maximum) indicates that they probably would have survived had dosing ceased. The ontogenetic delay is therefore probably a sublethal effect. This dosing and then cessation of dosing would resemble the field condition of heavy contamination for several weeks after application of a pesticide.
The delaying effect of dieldrin appears to be at work during the entire ontogeny, not just at one step. If dieldrin delayed only one step, say the approach, then once an approach was initiated the steps following the approach would have occurred after the elapse of the same time intervals as the control group. That is, the section of the ontogeny following the approach would be delayed as a unit. This was not the case. Approach was delayed 2.8 days, wing spread 3.7 days, attack 4.1 days, kill 5.1 days, and grab 7.1 days.
Mechanisms involved in pesticide-induced behavioral changes are unknown, but nerve tissue appears to be involved. O'Brien (1967: 16) stated that "in virtually every case where we understand the basis of insecticidal action, the effects are directly upon nerve." Matsumura and Hayashi's (1966) work suggests that dieldrin affects axionic transmission by complexing with neural membranes. They found that dieldrin has a much lower affinity for nerves of dieldrin-resistant cockroaches than for nerves of nonresistant cockroaches, and that dieldrin, once in contact with nerve tissue, cannot be removed by many solvents.
Whatever the explanation of the dieldrin-induced delay in the ontogeny of mouse killing, the delay could adversely affect the ability of treated shrikes to exploit effectively a readily-available food source that can be obtained with a small energy expenditure per unit of energy gained. But even if this 5-day delay in mouse killing is unimportant in survival of young shrikes, its existence indicates that pesticides are capable of inducing a subtle and unexpected behavioral change.
This paper is based on a portion of a master's thesis submitted to San Diego State University. I thank Ronald F. Monroe, Gerald Collier, Robert C. Harrison, and Roger F. Carpenter for reading my thesis and making helpful suggestions for revising it. I also thank Marcia Moriarty for her help in preparing the manuscript.
Anderson, J. M., & M. R. Peterson. 1969. DDT: Sublethal effects on brook trout nervous system. Science 164: 440-441.
Basson, N. C. J. 1971. Effect of dieldrin and its photoisomerization product "photodieldrin" on birds. Phytophylactica 3: 115-124.
Baxter, W. L., R. L Linder, & R. B. Dahlgren. 1969. Dieldrin effects in two generations of penned hen pheasants. J. Wildl. Mgmt. 33: 96-102.
Busbee, E. L. 1976. The ontogeny of cricket killing and mouse killing in Loggerhead Shrikes (Lanius ludovicianus L.). Condor, 78: 357-365.
Genelly, R. E., & R. L. Rudd. 1956. Chronic toxicity of DDT, toxaphene, and dieldrin to ringnecked pheasants. California Fish and Game 42: 5-14.
Gowdey, C. W., A. R. Graham, J. J. Sequin, & G. W. Stavraky. 1954. The pharmacological properties of the insecticide dieldrin. Canadian J. Biochem. Physiol. 32: 498-502.
Graber, R. R., S. L. Wunderlee, & W. N. Bruce. 1965. Effects of a low-level dieldrin application on a Red-winged Blackbird population. Wilson Bull. 77: 168-174.
Hansen, D. J. 1969. Behavior of estuarine organisms. Quarterly Report, Jun. 1969, of Center for Estuarine and Menhaden Research. Beaufort, S. C., Bur. Com. Fish.
Heath, R. G., & L. F. Stickel. 1965. Protocol for testing the acute and relative toxicity of pesticides to penned birds. Pp. IS 24 in The effects of pesticides on fish and wildlife. U.S. Fish Wildl. Serv. Circ. 226.
Jefferies, D. J., & B. N. K. Davis, 1968. Dynamics of dieldrin in soil, earthworms, and Song Thrushes. J. Wildl. Mgmt. 32: 441-456.
Kenaga, E. E. 1973. Factors to be considered in the evaluation of the toxicity of pesticides to birds in their environment. Environmental Quality and Safety 2: 166-181.
Korschgen, L. J. 1970. Soil-food-chain-pesticide wildlife relationships in aldrin-treated fields. J. Wildl. Mgmt. 34: 186-199.
Matsumura, F., & M. Hayashi. 1966. Dieldrin: Interaction with nerve components of cockroaches. Science 153: 757-758.
McEwen, L. C., & R. L. Brown. 1966. Acute toxicity of dieldrin and malathion to wild Sharp-tailed Grouse. J. Wildl. Mgmt. 30: 604-611.
Miller, A. H. 1931. Systematic revision and natural history of the American shrikes (Lanius). Univ. California Pub. Zool. 38: 11-242.
O'Brien, R. D. 1967. Insecticides: Action and metabolism. New York, Academic Press.
Ogilvie, D. M., & J. M. Anderson. 1965. Effect of DDT on temperature selection b%. young Atlantic salmon, Salmo salar. J. Fish. Res. Board Canada. 22: 503-512.
Sandler, R. E., G. A. Van Gelder, D. D. Elsberry, W. B. Buck, & G. G. Karas. 1969. Dieldrin exposure and vigilance behavior in sheep. Psychonomic Sci. 15: 261-262.
Sobotka, T. J. 1971. Behavioral effects of low doses of DDT. Proc. Soc. Exp. Biol. Med. 137: 952-955.>
Stickel, W. H., L. F. Stickel, & J. W. Spann. 1969. Tissue residues of dieldrin in relation to mortality in birds and mammals. Pp. 174-204 in Chemical fallout-current research on persistent pesticides (M. W. Miller and G. G. Berg, Eds.). Springfield, Illinois, Charles C Thomas.
Tucker, R. K. & D. G. Crabtree. 1970. Handbook of toxicity of pesticides to wildlife. U.S. Fish Wildl. Serv., Bur. Sport Fish Wildl., Res. Publ. No. 84.
Gelder, G. A., W. B. Buck, R. E. Sandler, J. Maland, G. G. Karas, & D. D. Elsberry. 1969. The effects of dieldrin and ruelene exposure on experimental behavior and the electroencephalogram. Pp. 125~133 in The biological impact of pesticides in the environment. Environ. Health Ser. 1. Corvallis, Oregon State Univ.
Wooley, D. E. 1969. Effects of DDT on the nervous system of the rat. Pp. 114-124 in The biological impact of pesticides in the environment. Environ. Health Serv. 1. Corvallis, Oregon State Univ.
The Effects of Dieldren on the Behavior of Young Loggerhead Shrikes